Explain Molarity Equation with its Limitations
Molarity Equation
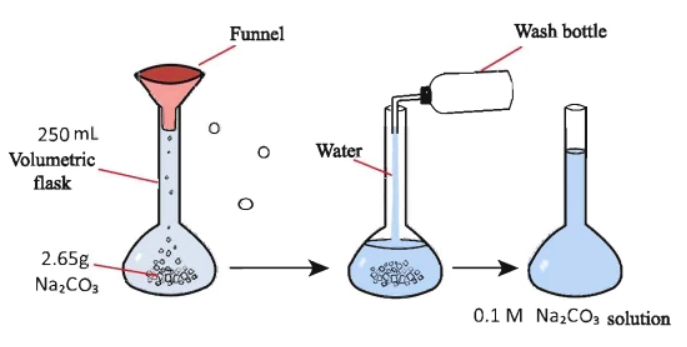
The mathematical formula used to estimate a solution’s molarity [M] is called Molarity Equation.
Molarity Formula
The formula is:
Molarity [M] = moles of solute [n] / volume of solution [V]
Where:
- Molarity (M) is the concentration of the solution in units of moles per liter (mol/L or M).
- Moles of solute (n) is the amount of solute present in the solution that is measured in moles.
- The volume of solution [V] is the solution’s total volume calculated in liters.
Any variable can be solved by using this equation. To calculate the number of moles of solute in a solution of known molarity and volume, the equation can be rearranged like
Moles of solute [n] = Molarity [M] x Volume of solution [V]
To calculate the volume of solution needed to dissolve a certain amount of solute to a desired molarity, the equation can be rearranged like…….
Continue to Read the Full Article
https://physicochemics.com/state-and-explain-molarity-with-its-formula/
